 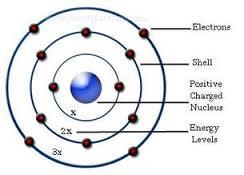 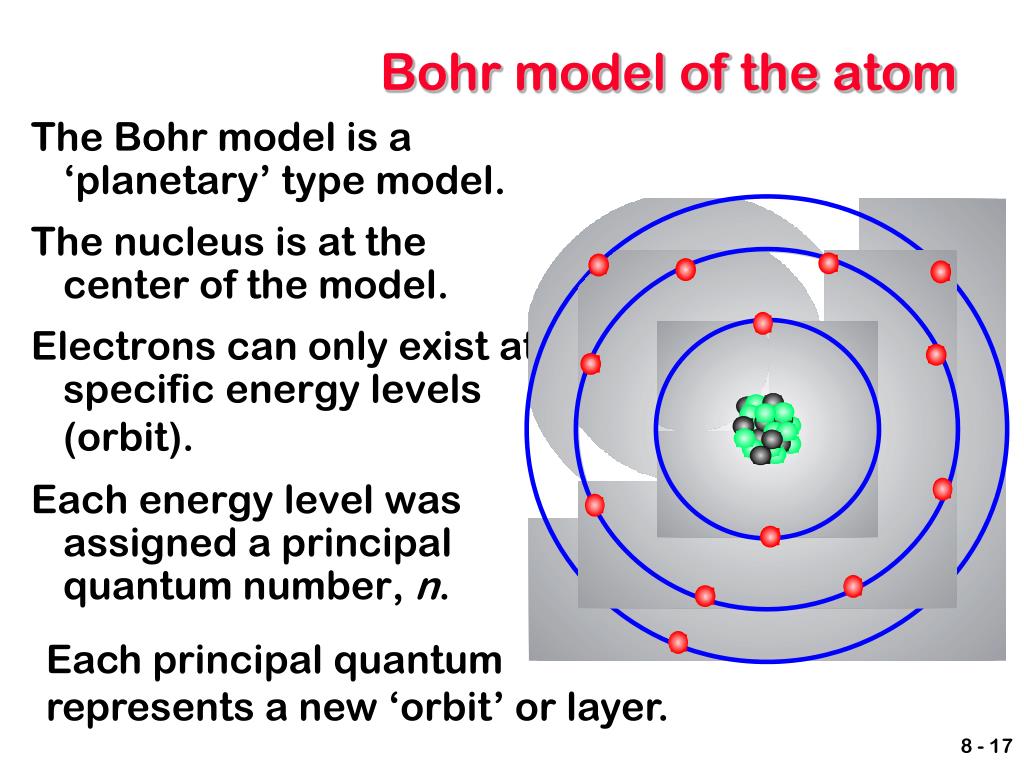
This behavior explains the discrete lines observed in atomic spectra.īohr’s atomic model is based on the postulates thatġ) The electron orbits the nucleus in fixed, circular paths called orbits or shells.Ģ) The electron’s energy is quantized, meaning that it can only exist in certain fixed amounts.ģ) The electron’s distance from the nucleus depends on its energy level.Ĥ) The number of electrons in an atom is fixed and is equal to the number of protons in the nucleus. When electrons transition between energy levels, they emit or absorb specific amounts of energy, resulting in the emission or absorption of light at distinct wavelengths. Emission Spectra: One of the significant contributions of the Bohr model was its ability to explain the atomic emission spectra observed in experiments.Conversely, when an electron loses energy, it moves to a lower energy level (ground state) and emits energy in the form of electromagnetic radiation. When an electron gains energy, it moves to a higher energy level (excited state). Energy Transitions: Electrons can transition between energy levels by either absorbing or emitting energy.These stable orbits are referred to as stationary states or stationary orbits. Stationary Orbits: Bohr suggested that electrons move in circular orbits around the nucleus but can only occupy certain stable orbits without emitting or absorbing energy.Electrons in the lowest energy level (n = 1) have the lowest energy, while those in higher energy levels have progressively higher energy. Quantized Energy: Bohr proposed that electrons can only exist in certain discrete energy levels and not in between. 
The energy levels are represented by whole numbers (n = 1, 2, 3, …) and are arranged at increasing distances from the nucleus. Each energy level is associated with a fixed amount of energy. Energy Levels: According to Bohr’s model, electrons orbit the nucleus in specific energy levels or shells. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
